Certificates
Glutathione
Current: LOT # ES-GTT-15K-B1
Epitalon
Current: LOT # ESEPI4350B1
Tesamorelin
Current: LOT # ES-TS-10-B1
ES-31 (SS-31)
Current: LOT # ES2S4350B1
Cagrilintide
Current: LOT # ES-CAG-10-CL1
Thymosin Alpha-1
Coming Soon
The Eden Scientific Standard
We believe in quality, for you and your research. To us, quality is a system – designed to demonstrate ownership and responsibility of our products.
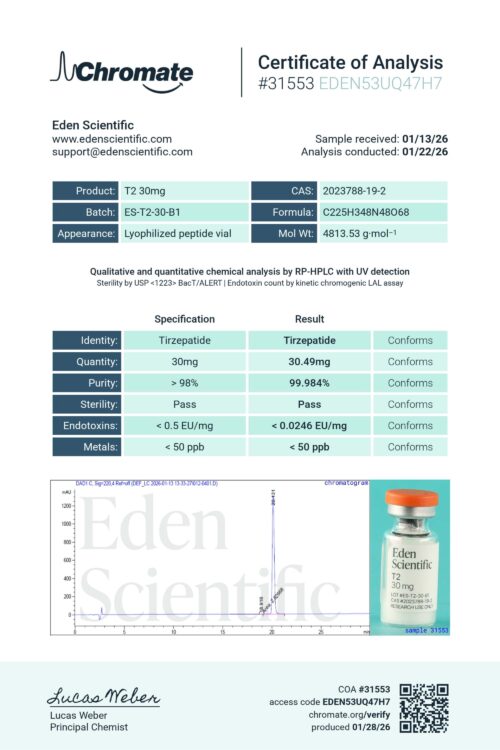
Every Eden product are third-party tested using USP (United States Pharmacopeia) aligned methods by independent laboratories.
Below is how the Eden Standard system works, explained with clarity:
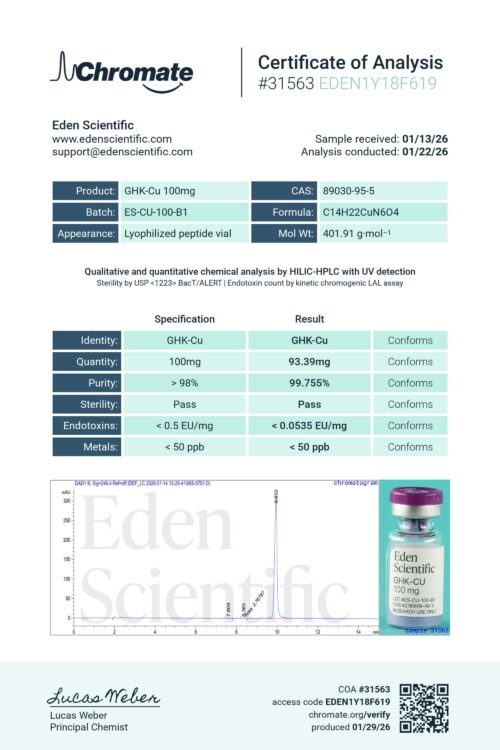
1. Identity Analysis
What it means
Identification confirms that each compound is exactly what it claims to be.
Why it matters
Misidentification is one of the most common causes of downstream variability. If the identity isn’t correct at the start, no amount of work can fix this.
How it’s tested
Identity is confirmed using RP-HPLC with UV detection, a precise analytical method that separates and quantifies components to confirm the expected composition.
2. Purity Analysis
What it means
Purity analysis measures the purity (how much of the material is the intended compound) expressed as a percentage.
Why it matters
Impurity affects consistency, introduces noise, and compromises results.
How it’s tested
Purity is confirmed using RP-HPLC with UV detection, a precise analytical method that separates and quantifies components to confirm the expected composition.
3. Net Content Analysis
What it means
Net content confirms that the amount provided matches what’s stated on the label.
Why it matters
Consistency depends not only on composition, but on quantity. If the stated amount isn’t reliable, results become harder to interpret and replicate.
How it’s tested
Net content is confirmed using RP-HPLC with UV detection to verify alignment between labeled amount and actual material.
4. Endotoxin (LPS) Analysis
What it means
Endotoxin screening checks for bacterial byproducts (Gram-negative bacteria) such as lipopolysaccharides (LPS) that can remain even when no live microorganisms are present.
Why it matters
Endotoxins are invisible and resistant to standard sterilization. If left unchecked, they can cause dangerous outcomes in research applications.
How it’s tested
Standard gel-clot Limulus Amebocyte Lysate (LAL) test to detect bacterial endotoxins. Results are reported as pass/fail based on a defined sensitivity threshold.
5. Sterility Analysis
What it means
Sterility (bioburden) screening evaluates whether unwanted levels of microorganism growth is present.
Why it matters
Microbial contamination introduces variables that can compromise consistency and reliability. Identifying this risk early protects downstream work.
How it’s tested
Sterility is tested using membrane filtration and incubation, screening for microbial contamination including bacteria, yeast, and mold. This qualitative test provides a pass/fail result indicating whether detectable microbial growth is present.
6. Heavy Metals Analysis
What it means
Heavy metals screening checks for trace elemental contaminants that don’t belong in research materials.
Why it matters
Because they aren’t detectable by appearance alone, heavy metals must be actively screened for.
How it’s tested
Heavy metals screening is conducted using colorimetric methods, allowing us to detect and assess the presence of unwanted elemental contaminants before release.